VA Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD)
A comprehensive, secure biorepository of specimens and associated data that affect Veterans health
About
Background

VA Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD) is a comprehensive, secure biorepository of specimens and associated data. These specimens and data are available to authorized VA investigators—and, under certain circumstances, to their external collaborators—to advance scientific understanding in support of developing diagnostic, therapeutic, and preventative strategies for use in clinical care.
VA SHIELD supports VA’s fourth mission: to improve the nation’s preparedness for national emergencies and to support emergency management, public health, safety, and homeland security efforts. As the largest integrated healthcare provider in the country and through its geographic breadth and size, VA is uniquely positioned to play a critical role in accelerating research efforts for future outbreaks. Through the establishment and implementation of the biorepository, VA is well-positioned to respond rapidly to national and international biological events and to substantially contribute to clinical research and development.
Vision and Purpose
Vision Statement: What is the goal of VA SHIELD operations?
Advancing rapid detection, therapy, and prevention of emerging diseases aimed at improving the quality and safety of health care for Veterans.
Purpose: What does VA SHIELD pursue?
Create a comprehensive infectious disease repository of specimens and associated data available to advance research, improve diagnostic and therapeutic capabilities, and develop strategies for immediate deployment in VA’s clinical environments.
Mission and Values
Mission: What does VA SHIELD do to achieve its goal?
The VA SHIELD biorepository provides researchers and clinicians with high-quality biosamples and comprehensive associated medical and sample data to accelerate the discovery-to-therapy pipeline.
Values: What values does VA SHIELD promote in its operations?
- Integrity: VA SHIELD maximizes the collection of high-quality specimens and data ethically and in compliance with applicable laws and regulations
- Synergy: VA SHIELD promotes cooperation with other VA programs, departments, governmental agencies, and private entities
- Quality: In its operations, VA SHIELD strives to adhere to the highest quality standards in the biorepository industry
- Efficiency: VA SHIELD provides an effective mechanism to collect, review, approve, and distribute specimens and data to scientific partners
Priorities and Defining Characteristics
VA SHIELD’s general priority is to develop a comprehensive repository providing access to researchers according to their needs and in line with VA ORD strategic priorities:
- Increase Veterans access to high-quality clinical trials
- Increase the substantial real-world impact of VA research
- Put VA data to work for Veterans
- Actively promote diversity, equity, and inclusion within our sphere of influence
- Build community through VA research
VA SHIELD considers the following priorities when granting specimen access:
- Scientific excellence: originality of the proposed work and perceived outcome and the ability to make an observable impact on the research subject
- Best use of specimens: identifying the proper use of biospecimens and ensuring, to the best of a researcher’s ability, that the specimens will be used for scientific excellence
- Harmonization/redundancy: ensuring investigators/researchers with overlapping goals and approaches collaborate on specimen usage
- Feasibility/impact on biorepository: ensuring the biorepository contains the type and quantity of specimens to support a request
- Validity/appropriateness: ensuring the type of biospecimens requested is the most appropriate to conduct the research proposed in addition to the proposed specimen size
- Funding: requestors/investigators with existing/conditional funding will have priority access to biospecimens
- Approval: evidence of IRB approval or operations project approval will be required before the release of biospecimens
- Specimen embargo: whether the specimens are on hold for a particular study
- Ethics: whether there are any ethical, human subject, or biohazard concerns
Defining Characteristics:
- Enterprise approach: VA SHIELD applies a strategic methodology to biospecimen collection and sharing across the VA Health Care System. We focus on streamlining and integrating business processes via communications, shared priorities, infrastructure and systems, capabilities, standardization, and improvement of the system. At VA SHIELD, we prioritize the success of the enterprise over individual success.
- Informational centralization: VA SHIELD sets Laboratory Information Management System (LIMS) as a single portal, a centralized electronic repository for collecting and storing biospecimen information.
- VA’s fourth mission: VA SHIELD’s policies, strategy, management, organizational, and operational structure is designed to support the Nation’s preparedness for national emergencies and to support emergency management, public health, safety, and homeland security efforts.
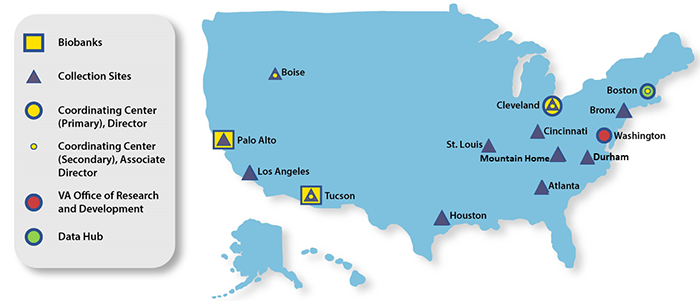
Locations
⇪




